| |
Computer Homeworks
Periodic Table
Consider that a file atomic.txt
contains a data of 103 different periodic elements. The first column is a atomic number, the second is
the name, the third is symbol and so on. Last two columns are melting point and boiling point in oC respectively.
Unknown property of an atom is assigned to a value of -1. Here is the part of the data file:
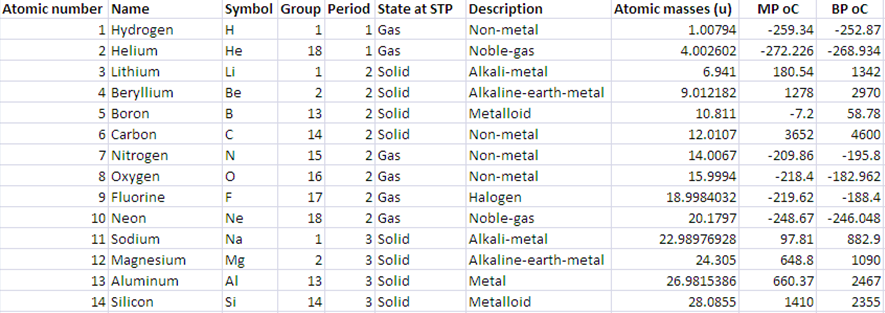
Write a C++ program that reads the data from the file into suitable vectors and then outputs a menu (given below)
to read an option from the keyboard:
******************************* MENU ***********************************
* *
* 0. Exit *
* 1. List properties of a specific atom *
* 2. List all properties of the elements belonging to a group *
* 3. List all properties of the elements belonging to a period *
* 4. List all properties of the elements for a given the description *
* 5. List all properties of the elements for a given the state at STP *
* 6. List all properties of the elements for a given boiling point range *
* 7. List all properties of the elements for a given melting point range *
* *
****************************************************************************
what is your choice? 1
- If the choice is 1 then program must read an atomic number (Z) from the keyboard and output all properties of the chosen element.
In this case, for Z = 1, the output of the program and its format should look like:
Input atomic number: 4
4 Beryllium Be 2 2 Solid Alkaline-earth-metal 9.0122 1278.0000 2970.0000
- If the choice is 2 then program must read a group number (G) from the keyboard and output all properties of the chosen group.
In this case, for G = 1, the output of the program and its format should look like:
Input group number: 1
1 Hydrogen H 1 1 Gas Non-metal 1.0079 -259.3400 -252.8700
3 Lithium Li 1 2 Solid Alkali-metal 6.9410 180.5400 1342.0000
11 Sodium Na 1 3 Solid Alkali-metal 22.9898 97.8100 882.9000
19 Potassium K 1 4 Solid Alkali-metal 39.0983 63.2500 759.9000
37 Rubidium Rb 1 5 Solid Alkali-metal 85.4678 38.8900 686.0000
55 Cesium Cs 1 6 Solid Alkali-metal 132.9055 28.4000 669.3000
87 Francium Fr 1 7 Solid Alkali-metal -1.0000 27.0000 677.0000
1 Hydrogen H 1 1 Gas Non-metal 1.0079 -259.3400 -252.8700
3 Lithium Li 1 2 Solid Alkali-metal 6.9410 180.5400 1342.0000
11 Sodium Na 1 3 Solid Alkali-metal 22.9898 97.8100 882.9000
19 Potassium K 1 4 Solid Alkali-metal 39.0983 63.2500 759.9000
37 Rubidium Rb 1 5 Solid Alkali-metal 85.4678 38.8900 686.0000
55 Cesium Cs 1 6 Solid Alkali-metal 132.9055 28.4000 669.3000
87 Francium Fr 1 7 Solid Alkali-metal -1.0000 27.0000 677.0000
- If the choice is 4 then program must read a description name.
In this case, for noble gases, the output of the program and its format should look like:
Input a description name: Noble-gas
2 Helium He 18 1 Gas Noble-gas 4.0026 -272.2260 -268.9340
10 Neon Ne 18 2 Gas Noble-gas 20.1797 -248.6700 -246.0480
18 Argon Ar 18 3 Gas Noble-gas 39.9480 -189.2000 -185.7000
36 Krypton Kr 18 4 Gas Noble-gas 83.7980 -156.6000 -152.3000
54 Xenon Xe 18 5 Gas Noble-gas 131.2930 -111.9000 -107.1000
86 Radon Rn 18 6 Gas Noble-gas -1.0000 -71.0000 -61.8000
- If the choice is 5 then program must read a state name.
In this case, for Gas, the output of the program and its format should look like:
Input state at STP: Gas
1 Hydrogen H 1 1 Gas Non-metal 1.0079 -259.3400 -252.8700
2 Helium He 18 1 Gas Noble-gas 4.0026 -272.2260 -268.9340
7 Nitrogen N 15 2 Gas Non-metal 14.0067 -209.8600 -195.8000
8 Oxygen O 16 2 Gas Non-metal 15.9994 -218.4000 -182.9620
9 Fluorine F 17 2 Gas Halogen 18.9984 -219.6200 -188.4000
10 Neon Ne 18 2 Gas Noble-gas 20.1797 -248.6700 -246.0480
17 Chlorine Cl 17 3 Gas Halogen 35.4530 -100.9800 -34.6000
18 Argon Ar 18 3 Gas Noble-gas 39.9480 -189.2000 -185.7000
36 Krypton Kr 18 4 Gas Noble-gas 83.7980 -156.6000 -152.3000
54 Xenon Xe 18 5 Gas Noble-gas 131.2930 -111.9000 -107.1000
86 Radon Rn 18 6 Gas Noble-gas -1.0000 -71.0000 -61.8000
- If the choice is 6 then program must read two boiling point values.
In this case, for [-200,+100], your program must output the elements whose boiling points are in the range [-200,+100] oC.
The output of the program and its format should look like:
Input a boiling point range: -200.0 +100.0
5 Boron B 13 2 Solid Metalloid 10.8110 -7.2000 58.7800
7 Nitrogen N 15 2 Gas Non-metal 14.0067 -209.8600 -195.8000
8 Oxygen O 16 2 Gas Non-metal 15.9994 -218.4000 -182.9620
9 Fluorine F 17 2 Gas Halogen 18.9984 -219.6200 -188.4000
17 Chlorine Cl 17 3 Gas Halogen 35.4530 -100.9800 -34.6000
18 Argon Ar 18 3 Gas Noble-gas 39.9480 -189.2000 -185.7000
36 Krypton Kr 18 4 Gas Noble-gas 83.7980 -156.6000 -152.3000
54 Xenon Xe 18 5 Gas Noble-gas 131.2930 -111.9000 -107.1000
86 Radon Rn 18 6 Gas Noble-gas -1.0000 -71.0000 -61.8000
91 Protactinium Pa 3 7 Solid Actinide 231.0359 1600.0000 -1.0000
96 Curium Cm 3 7 Solid Actinide -1.0000 1340.0000 -1.0000
97 Berkelium Bk 3 7 Solid Actinide -1.0000 -1.0000 -1.0000
98 Californium Cf 3 7 Solid Actinide -1.0000 -1.0000 -1.0000
99 Einsteinium Es 3 7 Solid Actinide -1.0000 -1.0000 -1.0000
100 Fermium Fm 3 7 Solid Actinide -1.0000 -1.0000 -1.0000
101 Mendelevium Md 3 7 Solid Actinide -1.0000 -1.0000 -1.0000
102 Nobelium No 3 7 Solid Actinide -1.0000 -1.0000 -1.0000
103 Lawrencium Lr 3 7 Solid Actinide -1.0000 -1.0000 -1.0000
|

