22.4.1999
EP 221 MATERIAL SCIENCE
FISRT MIDTERM
Time Duration : 110 Mins.
1.
Write
the important properties of engineering materials and then explain physical
properties. (8 points)
2.
a-)
Classify the atomic models. (5 points)
b-) Using Bohr’s postulates, derive an expression to find
the Bohr’s radius and total energy of an electron in the hydrogen atom. (20
points)
3. a-)
Define cation, anion,
electropositive and electronegative elements. (6 points)
b-) Classify the quantum numbers of electrons in atoms and
then explain principal quantum number (n). (8 points)
c-)
Write the electron
configuration of the elements 35Br, 46Pd, 14Sr+2,
29Cu. (6 points)
4.
a-) Classify
the types of the atomic and molecular bonding and explain covalent bonding in
detail. (10 points)
b-)
The attractive force
between ions with unlike charges is e2/4pe0r2
while the repulsive force may be written Ce2/rn where the
exponent n is about 10 and C is a constant. Obtain an expression for the
equilibrium distance (r0) between the ions in terms of C and n. (12
points)
5.
a-) Classify
the possible crystal structures in metals and give a brief information about
FCC. (5 points)
b-)
Explain atomic pacing
factor and then find it for SC, BCC and FCC. (20 points)
Good Luck Dr.A.N.YAZICI
21.5.1999
EP 221 MATERIAL SCIENCE
SECOND MIDTERM
Time Duration : 90 Mins.
1.
Give
the type of crystal imperfections in solids and then explain point defects in
detail. (20 points)
2.
Draw
the planes of Miller indices. (12 points)
(a) ![]() (b)
(b) ![]() (c )
(c ) ![]() (d)
(d) ![]() (e)
(e) ![]()
3. Draw the direction of the given Miller indices. (12
points)
(a) ![]() (b)
(b) ![]() (c )
(c ) ![]() (d)
(d) ![]() (e)
(e) ![]()
4. 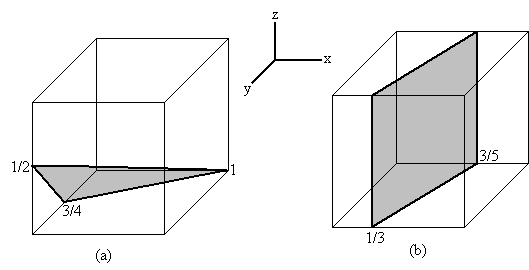 Determine the Miller indices of the cubic
crystallographic plane shown in below figures. (10 points)
Determine the Miller indices of the cubic
crystallographic plane shown in below figures. (10 points)
5. Copper has an atomic radius of 0.128 nm ( 1.28 Å), and
it has FCC crystal structure, the atomic weight of copper 63.5 gr/mole.
a-) Calculate the volume of copper in terms of atomic
radius r ? (5 points)
b-) Compute its density ? (6 points)
c-) Calculate the longest wavelength of X-rays which
will produce diffraction from the close packet planes. (15 points)
6. The diffusivity of Al in Copper is 2.5x10-17
m2/sec at 500 0C and 1.6x10-12 m2/sec
at 1000 0C.
a-) Determine the values of D0 and Q for
this diffusion couple. (15 points)
b-) What is the diffusivity at 750 0C. (5
points)
(NA=6.02x1023 atoms/mole R=8.314 J/mol.K 1
nm=1x10-9 m)
Dr.A.N.YAZICI
14.06.1999
EP 221 MATERIAL SCIENCE
Final Exam
Time Duration : 110 mins.
1.
Suppose that Fe2O3
has a density of 5.5 Mg/m3. How many atoms are present a-) Per mm3;
b-) Per gram ? (Fe = 56, O = 16) (10
points)
2.
What are the
forces acting between atoms and molecules? How do the mutual potential energies
of two atoms or molecules vary with their distance of separation ? (15 points)
3.
The density of FCC
gold is 19300 kg/m3 and its atomic weight is 197. Calculate the
separation between the close-packed planes. On the assumption that the atoms
may be thought as spheres which just touch one another, estimate the atomic
radius. (20 points)
4.
Draw the given
Miller indices in cubic crystal structure.
(15 points)
(a) ![]() (b)
(b) ![]() (c)
(c) ![]() (c )
(c ) ![]()
5.
Consider the gas
carburizing of a gear of 1018 stell (0.18 wt %) at 927 0C (1700 0F).
Calculate the time necessary to increase the carbon content to 0.25 wt % at
0.40 mm below the surface to be 1.00 wt % and that the normal carbon content of
the steel gear before carburizing is 0.18 wt %. {D (C in g-iron) at 927 0C) = 1.28 x 10-11
m2/sec}.Erf(Z) = 0.9103 when Z = 1.2 and Erf(Z) = 0.9340 when Z=
1.3 (20 points)
6.
A silver wire is
to conduct a 7 Amper current with a maximum voltage drop of 0.004 V/cm. What
must be the minimum diameter of the
wire in meters. At what temperature will the electrical resistivity of this
wire be 4.2 x 10-8 W-m. {s=6.3 x 107 (W-m)-1 at room temperature} (20 points)
Good Luck
Dr.A.N.YAZICI
21.4.2000
EP 221 MATERIAL SCIENCE
FIRST MIDTERM
Time Duration : 100 Mins.
1.
What
are the main differences between common engineering materials. (8 points)
2.
a-)
Classify the atomic models. (4 points)
b-) Explain Bohr’s atom model in detail. (10 points)
c-) Find the Bohr’s radius and total energy of last orbital
electron in iron ( 26Fe) atom. (6 points)
3.
a-) Calculate the required energy and wavelength of a
photon to jump an electron from n=1 to n=3 state in a Lithium (3Li)
atom. (8 points)
b-) Determine the shortest and
longest wavelengths of Balmer series of Hydrogen (1H) atom in
Angstrom (8 points).
4.
When two different
type of atoms A and B come together, they form one type of bond between them.
Decide the type of bonding between them and then calculate the minimum bond
energy according to the given values. (18 points)
a-) UA-A=
4 eV, XA= 2 eV, UB-B=9
eV, XB=2 eV
b-) UA-A=
4 eV, XA= 2 eV, UB-B=9
eV, XB=2 meV
5.
Find the APF of BCC and HCP structure. (Assume that c=1.63a in HCP). (20
points)
6. a-)
Draw the direction of the given Miller indices in cubic crystal structure. (9
points)
(i) ![]() (ii)
(ii) ![]() (iii )
(iii ) ![]()
b-)
Determine the Miller indices of the cubic direction between the position
co-ordinates ![]() and
and ![]() , then draw it in a unit cell. (9 points)
, then draw it in a unit cell. (9 points)
Useful Constant
ħ=1.05x10-34 J/sec (ħ=h/2p) e=1.6x10-19
C 1A0=1x10-10
m
k=9x109 N/C R=1.097x10-3
A-1![]()
me=9.1x10-31 kg 1 eV=1.6x10-19
J